 |
Membrane Pharmacy Structure DynamicsResearch group : Priv.Doz. Dr. Thomas NawrothStructural Biology |
 |
Membrane Pharmacy Structure DynamicsResearch group : Priv.Doz. Dr. Thomas NawrothStructural Biology |
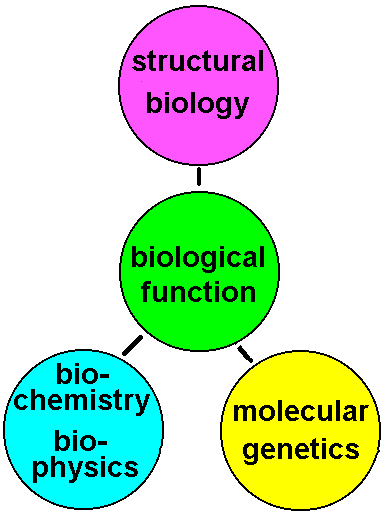 |
Fig.1: The description and manipulation of biological function requires three regimes of life sciences: Structural biology, Molecular genetics and Biochemistry or Biophysics (i.e. function analysis / enzymatics, isolation and purification of systems and protein chemistry) |
|
|
|
|
|
|
|
|
| X-ray diffraction with monochromatic beam | indirect | 0.2 - 0.5 nm | single molecule (20 nm) | yes, 3D- crystals of highly purified, structural homogenous protein, 100 mg material | well ordered 3D- crystals and metal derivatives or anomaouls scatterers incorporated (MAD phasing) | restricted (flash photolysis), at high flux synchrotorns, e.g. ESRF - ID14 beamlines; ELETTRA - 5.1 beamline |
| X-ray Laue-diffraction with polychromatic beam | indirect | 0.2 - 0.5 nm | single molecule (10 nm) | yes, 3D- crystals of highly purified, structural homogenous protein, 100 mg material | well ordered 3D- crystals and derivatives required; restricted to small proteins of high scattering power | yes, e.g. ESRF - ID beamline |
| SAXS, small amgle X-ray scattering;
ASAXS, anomalous small amgle X-ray scattering |
no, only by molecular modelling | 1 nm | single particle or cluster (500 nm) | yes, solution of highly purified, structural homogenous protein, 1-10 mg material | no, but purified, structural homogenous protein required;
domain differentiation in complexes by metal labeling possible |
yes, TR-SAXS by rapid mixing (stopped flow technique) or flash photolysis (caged compounds, chromoproteins) at high flux synchrotrons, e.g. ESRF - ID2A, ID1, ID13 beamlines; ELETTRA - SAXS (5.2) |
| SANS, small amgle neutron scattering | no, only by molecular modelling | 1 nm | single particle or cluster, membranes (2000 nm) | yes, solution of highly purified, structural homogenous protein or membranes, 5-50 mg material | no, but purified, structural homogenous protein required;
domain differentiation in complexes by specific deuteration or spin labeling possible |
yes, TR-SANS at high flux neutron sources, e.g. ILL - D22 , D11 beamlines |
| inelastic and quasielastic neutron scattering, TOF | no, only by molecular modelling | (1 nm) | single domains in proteins or clusters, membranes | yes, solution or crystal of highly purified, structural homogenous protein or membranes, 10-100 mg material | no, but purified, structural homogenous material required;
domain differentiation in complexes by specific deuteration or spin labeling possible |
yes, TOF experiments at high flux neutron sources, e.g. ILL - D22 and several other beamlines |
| Neutron diffraction | indirect | 0.3 - 0.5 nm | single molecule (20 nm) | yes, 3D- crystals of highly purified, structural homogenous protein, some 100 mg material | well ordered 3D- crystals and deuterium derivative(contrast phasing) | no; method available only at high flux neutron sources, e.g. ILL |
| EXAFS and XANES | indirect, only by molecular modelling | 0.001 nm | 0.5 nm, environment of metal center | restricted, some 100 mg material | sample should be homogenous in metal centers | yes, at high flux synchrotorns, e.g. at ESRF - beamlines ID26 (scanning), ID24 (energy dispersive) |
| 2D/3D- NMR | indirect from NOE's and angles | 0.4 nm | single molecule (10 nm) | yes, solution of highly purified, structural homogenous protein, 100 mg material | 13C- and 15N-derivatives required; restricted to small proteins (< 40,000 mass with 800 MHz-NMR) | no, until now only by simulation of protein flexibility |
| FT-IR | no, only by molecular modelling | only in labeled domains | single molecule (20 nm) | yes, solution of highly purified, structural homogenous protein, 100 mg material | 13C- derivatives required; site specific isotope labeling | yes |
| microscopy (light) | yes | 500 nm ( = light wavelength) | micrometer | no | thin solid sample / cut | yes, video microscopy |
| fluorescence microscopy (light) | yes | < 500 nm (light wavelength) | micrometer | no | fluorescence labeling, thin solid sample / cut | yes, video microscopy |
| laser scanning microscopy | yes | 150 nm ( = 1/4 wavelength) | micrometer | no | thin solid sample / cut | restricted |
| confocal laser microscopy | indirect, modelling | 1-2 nm (chromophore) | single molecule (20 nm) | yes | single molecule is held in the focus of a laser, radiation demage risk | yes |
| electron microscopy, negative stain technique | yes | 2 - 3 nm | 0,1 - 10 micrometer | no | stain with heavy element, may produce artefacts with membranes and flexible proteins | no |
| electron microscopy, freeze fracture technique | yes, indirect | 2 - 3 nm | 0,1 - 10 micrometer | no | rapid freezing (vitrification) and subsequent cut and metal sputtering in vacuuum | restricted, by time resolved freezing of the samples |
| electron diffraction (2D-crystallography) | indirect | 0.3 - 1 nm | single molecule (20 nm) | yes, 2D- membrane protein crystals | 2D- crystals of vaccum stable sample derivative required | no |
| AFM , atomic force microscopy | indirect | 1-2 nm | 0,1 - 10 micrometer | no | solid sample with rigid surface required | no |
| Photoaffinity labeling, Photoaffinity crosslinking | no | 0,5 nm group distance | 1-5 nm in single particle or cluster, membranes | recommended for unambigous results | molecule must have a few reactive groups at surface | yes, but not usual; by flash photochemistry |
| MD , molecular dynamics structure simulation | by theory | < 0.1 nm | single molecule (10 nm) | - (theory) | restricted to small moleules and short times (picoseconds) | yes, may be improved, if more experimental data on molecular motion become available |