 |
Membrane Pharmacy Structure Dynamics
Research group : Priv.Doz. Dr.
Thomas Nawroth
Technology
|
Topics: Overview, Protein
purification and membrane preparation, Chemical
synthesis of caged compounds and metal labels, Optics
and flash technology, Rapid mixing and stopped
flow technology, Sample environments for spectroscopy
, X-ray and neutron experiments, Computer
control and electronics
Overview
[ Impressum-Disclaimer
]
For the investigation of molecular motions in proteins, membranes and artificial
systems the MPSD group follows an interdisciplinary
concept. This combination of complementary methods is applied at the
scientific level and at the technological side.
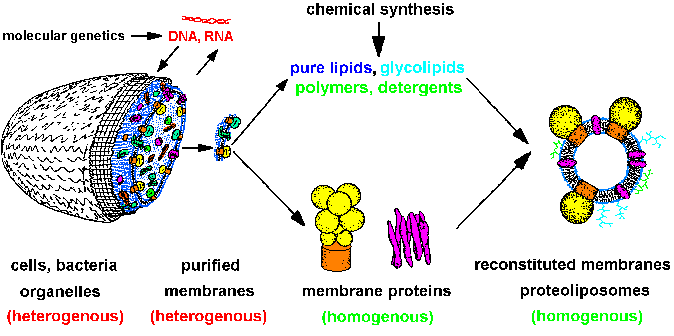
|
| Fig.1: The strategy of structure
and membrane research leads from heterogenous cells over isolated homogenous
systems (proteins, lipids, membranes, nucleic acids) to reconstituted model
membranes and protein-detergent complexes (after T20).
Molecular genetics supplies cells with specific proteins, chemical synthesis
gives us lipids, polymers and compounds for optical system manipulation,
e.g. caged-compounds. The molecular homogenous systems are suitable for
structure investigation (SAXS, SANS, ASAXS, X-ray diffraction, EXAFS /
XANES, especially with time resolution). |
The supply of the biological systems require the growth of cells from
a genetically defined strain and subsequently the isolation
and purification of proteins in the native state. For investigation
of the structure-function relation and molecular biochemistry, this means
isolation of milligram-amounts of pure proteins (e.g. 99%) in a functional
competent and homogenous state. While working with proteins
of the bioenergetic system, this includes the preparation and purification
of native membranes, e.g. protoplast membrane vesicles (PMV) from Micrococcus
luteus, or membranes from cell organelles (mitochondria). During the
study artificial
model membranes have to be
prepared, e.g. liposomes, which are sufficient for function and structure
investigation, e.g. with neutron scattering. The technology for supplying
the biological system is presented in the
protein
preparation page.
Some scientific investigations require the chemical synthesis of several
small molecules for the manipulation of the system or the generation of
a specific signal during structure investigation. In the MPSD group those
synthesis work is done at two compound classes: i) the synthesis of caged
compounds (caged-ATP, caged-nucleotides, caged-tritium, caged acids / proton;
ref. T7, T9,
T14,
T15
, T20) and ii) the synthesis of structure
labels, i.e. artificial metal labels for proteins, lipids and polymers,
e.g. Europium-chelates, and fluorescent or coloured photoaffinity labels
(e.g. the hydrophobic labels in ref. F15).
An overview and some receipts are given in the Chemical
synthesis chapter.

|
Fig.2: A 4-beam dual-wavelength
flash illumination device for the manipulation of proteins and membranes
by light. The sample (e.g. reconstituted Bacteriorhodopsin/ ATP-synthase
liposomes) is illuminated homogenously by two opposite beams of visible
light (green, 550 +- 40 nm; 1000 mW/cm2). After a reaction delay the sample
is flash-irradiated by two lateral UV sources (300-380 nm; 80 J) for photolabeling
or activation of caged compounds. Analogous devices are used for time resolved
X-ray and neutron scattering with caged compounds (caged-acid/ proton)
(after T20). |
A variety of experiments about enzymatic analysis, surface labeling,
structure research with X-rays and neutrons, and generation of functional
states in time resolved studies requires a sofisticated optical setup.
Especially experiments with photogeneration of compounds, e.g. for light-driven
ATP-synthesis, cleavage of caged compounds and photoaffinity labeling or
crosslinkig, yield unambigous results only if the optical system is setup
properly, i.e. if the sample is homogenously illuminated at any point.
Similar precautions have to be observed, if the status of the system is
examined by optics, e.g. fi probing the protein conformation by a fluorescence
measurement. For triggering of a time resolved experiments by light, e.g.
photolysis of a caged acid or caged ATP, the light has to be generated
in a short time (< 1 ms) and large amount (several J/ml sample). For
this purpose the MPSD groups uses currently mainly (UV) flash illumination.
The technologies are presented in the optics page.
If the molecular reaction (motion) is slower than a few milliseconds
and if the system is insensitive to shearing forces, it can be investigated
in a more simple way after triggering by a concentration jump during rapid
mixing of the compounds with a stopped flow
device. The MPSD group has constructed for this purpose several devices
and sample environments since 1987 (ref. F14,
T14,
T20)
and improved the technology for the use at high flux synchrotrons (ref
, S##, F##) and time resolved neutron scattering (R##). A result is shown
in the structural film of working F1ATPase.
The mixing technology is demonstarted in the rapid
mixing page.

|
Fig.3: A flow-through quartz capillary
for structure investigation of protein solutions as used at DESY-HASYLAB,
Hamburg, at the JUSIFA beamline (B1). Analogous, but more complicated
sample environments with helium-jet cooling and stopped-flow mixing devices
or flash units are used for time resolved studies at the high flux synchrotrons
(ESRF, ELETTRA)
and for time resolved neutron scattering at the ILL,
Grenoble. |
X-ray and neutron scattering experiments of biological systems require
special sample environments, if examined with high flux, high precision
and / or time resolution. Thus the MPSD group has in cooperation with industry
constructed in the last 20 years several sample
environments for structure research of biomolecules in solution with
X-ray synchrotron radiation, neutrons and spectroscopy. A sketch of an
recent helium cooled sample environment SBox4 for high flux synchrotron
light scattering is shown in Fig.4 and ref. S##. The principal setup of
a typical X-ray setup is shown in Fig.5. This actual environment, which
is suitable also for temperature jump (cold trap) experiments, is presented
in the sample environment page.
The control of time resolved experiments
and data aquisition is done at the MPSD group by a combination
of computer- and digital eletronics (TTL). During this several novel
digital and electronics divices were constructed in cooperation with industry.
The first experiments in the early 80's where done with timer-chains. Since
1988 we switched over to microcomputer systems (with directly coded binary
code for hardware controller CPU's) and TTL-electronics for the fast process
steps (microseconds to milliseconds) and PC-computer programs for the slower
steps (> 100 ms), providing the parameter and data aquisition / handshake
(e.g. F14,
T14,
T20).
An actual example is given in the WEB for time resolved neutron scattering
of liposomes (http://www.ill.fr ; see experiment
reports ... database-settings: instrument = D22 ; name = Nawroth ; experiment
= Test-251 and further). Some technological aspects are presented in the
computer
control and electronics page.
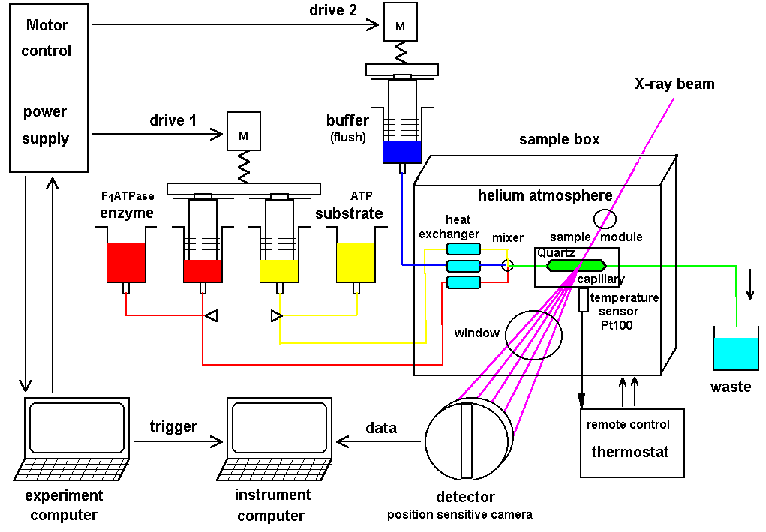
|
| Fig.5: Principal setup for a time
resolved X-ray scattering and absorption experiment at a high flux
synchrotron. The active working sample is produced from stock solutions
by rapid mixing with a stopped-flow device.
The helium atmosphere reduces air scattering and beam heating. Radiation
demage can be suppressed by slow continous sample displacement by drag
buffer with a second drive (from R6, Fig.1). |
email to: nawroth@MPSD.de
update : 15.10.2013






