 |
Membrane Pharmacy Structure DynamicsResearch group : Priv.Doz. Dr. Thomas NawrothSample Environments for Structure Research |
 |
Membrane Pharmacy Structure DynamicsResearch group : Priv.Doz. Dr. Thomas NawrothSample Environments for Structure Research |
Topics: sample cells, capillary, flat cell, environments, stopped-flow / flash and temperature jump (cold trap double jump), neutron scattering
X-ray and neutron scattering experiments of biological systems require special sample environments, if examined with high flux, high precision and / or time resolution. Thus the MPSD group has (in part in cooperation with industry) constructed in the last 20 years several sample cells and environments for structure research of biomolecules in solution with X-ray synchrotron radiation, neutrons and spectroscopy.
The optimal sample cell type for structural biology experiments depends on the type of experiment and the dimensions and stability of the used beam (X-eay, light, neutrons). According to table1, a capillary is the cell of choice for experiments including a presure jump, e.g. stopped-flow investigations, and for studies usind a narrow, stable beam (< 1mm). With lager beams (neutrons), lower energy (X-ray < 6 keV) and samples sensitive to shearing forces a flat window cell has to be choosen. In this case a time resolved stuy requires flash photochemistry as trigger.
Table1: Comparison of some sample cell types for structural biology experiments with fluids.
| cell type | advantages | disadvantages | window materials |
| capillary, one side open (Mark capillary) | easy to use,
cheapest cell precision sufficient for crystallography |
low reproducibility of background,
low-Q limit for SAXS / ASAXS no direct use in vacuum surface reflection of slit scattering |
glas, quartz |
| capillary, flow through | high reproducibility as required for SAXS (3*10e-5 I/Io possible)
withstands pressure jumps (> 30 bar) |
expensive,
difficult tubing (active sodium surface etching of PTFE required) low-Q limit for SAXS / ASAXS surface reflection of slit scattering |
quartz |
| foil cell, fixed ("Stuhrmann cells") | easy to use
no surface reflection |
low reproducibility of pathlength,
variable Capton diffraction ring difficult to fill with detergent solution background/signal ratio varies |
Mylar, Mica, Nalophane, Capton |
| flat window cell, flow-through;
in vacuum or at low energy with 4 windows and Helium flushing |
high reproducibility for ASAXS and EXAFS (10e-6 I/Io possible)
homogenous illumination by additional light/flash beams possible temperature constant with He-cooling no surface reflection constant background/signal ratio adjustabvle pathlength down to 25 µm large beam possible (< 6 mm) |
special flat window cell required,
large cell housing (60x80 mm) windows have to replaced daily at high flux synchrotron (10e13 ph/s at sample) and low energy (< 8 keV) |
Mylar, Mica, Nalophane
in vacuum: double windows (i.e.: 4) with helium flushing |
Table2: Samples cells used by the MPSD group for structural biology experiments.
| cell code , [photo] | cell type | sample exchange / fill | comments |
| capillary cells : | |||
| CC1 | quartz capillary, 1 side open, flushed | static / syringe with long needle | tests at DESY-HASYLAB |
| CC2 | quartz capillary on aluminium support, beam window 15x3 mm; 1.1 mm diameter | flow-through with PTFE tubing | used at DESY-HASYLAB beamline A1 and B1 in vacuum, and in the frist experiments at ELETTRA |
| CC3 | quartz capillary on aluminium support, window 30x15 mm for helium-jet cooling; 1.3 mm diameter | flow through with PTFE tubing | used at ESRF, ELETTRA and DESY-HASYLAB beamline B1 (JUSIFA) |
| flat window cells : | |||
| FC1 | flat cell with 2 mylar windows and o-ring, pathlength differs | static, fill during assembly | "Stuhrmann cells" used at DESY-HASYLAB beamline A1 |
| FC2 | flat window flow-trough cell for spectroscopy with 4 mylar windows, 10 mm windows, helium flushed | flow-through with PTFE tubing, thermostatisation in cell body | for spectroscopy and photochemistry experiments |
| FC3 | flat window flow-trough cell for ASAXS/ EXAFS with 4 mylar windows, 3 mm windows, helium flushed | flow-through with PTFE tubing, narrow pathlength (> 25 µm) for
low energy X-ray experiments (U, S, P);
pathlength constant, adjustable |
used at DESY-HASYLAB beamline A1 for ASAXS experiments |
| FC4 | flat window flow-trough cell for ASAXS/ EXAFS with 4 Nalophane windows, 8 mm windows, helium flushed, equipped for additional flash / optics beams (dual beam experiments) | flow-through with PTFE tubing, pathlength constant, adjustable | used for SAXS, ASXAS, EXAFS and flash photolysis experiments at ESRF (ID1, ID2A, ID24) and DESY-HASYLAB (B1, BW4) |
 |
 |
| Fig.1: A flow-through capillary unit (CC2) is equipped with etched PTFE tubing on a alumminium support (window 3 x 15 mm). | Fig.2: A vacuum-tubing plug is required for sample exchange without interuption of the vaccum and time resolved experiments. |
The evaluation of SAXS and ASAXS experiments with protein and polymer
solutions requires four data sets, which have to be estimated under identical
conditions: scattering and transmission of i) empty cell; ii) the
solvent filled cell, iii) the macromolecule solution in the cell, and iiii)
a dark exposure (T(sample) = 0). Due to our experience this is with capillaries
only possible with the required precision (10e-4 to 10e-5 with 1% protein)
with a flow-through capillary cell. Furthermore the beam hight has to be
smaller than the diameter of the capillary and stable in position, at least
in the vertical direction (see below, fig.4). Thus the MPSD group uses
flow-trough cells, even if static experiments are done (non time resolved).
Fig.1 shows a flow-through quartz capillary (1.3 mm diameter, Hilgenfeld,
Germany), which has been equipped with teflon tubing (0.8 mm i.D., as usual
for HPLC / FPLC) and fixed on an aluminium support (beam window 3 x 10
mm). The outer surface of the PTFE tubing ends (20 mm) had been etched
with an active sodium suspension prior to fixation with 2-component resin
(ChemGrip PTFE kit from Sigma) (Note: be careful with that, it is self-inflammable
and extremely caustic). After equipment with a vacuum-tubing plug (Fig.2)
this type of flow-through can be used in vacuum. The refill does not require
an interuption of the vacuum. Those setups, as shown in Fig.3, have been
used at the B1 (JUSIFA) and A2 beamlines at DESY-HASYLAB for static and
time resolved SAXS experiments and for EXAFS experiments with dilute solutions
using the "true reference method" (Fig.4 from R##).
 |
Fig.3: A flow-through quartz capillary (CC3) for structure investigation of protein solutions as used at DESY-HASYLAB, Hamburg, at the JUSIFA beamline (B1). Analogous, but more complicated sample environments with helium-jet cooling and stopped-flow mixing devices or flash units are used for time resolved studies at the high flux synchrotrons (ESRF, ELETTRA) and for time resolved neutron scattering at the ILL, Grenoble. |
 |
Fig.4: Setup for SAXS, ASAXS and corresponding EXAFS experiments with a flow-through cell using the "true reference method" for structure investigation of protein and metal label solutions as used at the JUSIFA beamline (B1), DESY-HASYLAB, Hamburg, (from S38). |
With any capillary a special cross-like background signal with a vertical
extension is observed at beamlines which use slits, i.e. after beanding
magnets and wiggler sources. By experiments at JUSIFA, ELETTRA and ESRF-ID2A
we found the origin, at least for our capillaries: As depicted in Fig.5,
the signal is generated by total reflection of the slit scattering of the
beam definition slit (S2) at the opposite surface of the capillary. Thus
it cannot be abolished by closing the guard slit (S3) between S2 and the
cell (only if the beam height is definitely zero). Consequently the signal
was absent during time resolved experiments at ESRF - ID2A, where we used
a double undulator (point source) setup with all slits fully open (focus
at sample: 0.2 x 0.8 mm; 1.5*10e13 ph/s at 12.5 keV).
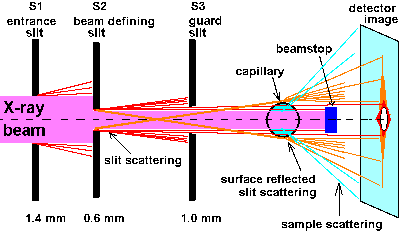 |
| Fig.5: The vertical extension of the cross-like background signal from capillary cells in bending magnet and wiggler beamlines is generated by surface total reflection of the slit scattering from the beam definition slit (S2) at the opposite capillary side (slit names as at JUSIFA beamline (B1), DESY-HASYLAB, Hamburg). |
 |
|
| Fig.6: Fluid samples can be subjected to X-ray and spectroscopic investigations in a vaccum for some hours using simple closed flat cells with polymer windows ("Stuhrmann cells", FC1). The sketch (a) shows the assembly from an aluminium support, 1st polymer-window (6 µm Mylar), O-ring (12x 1 mm), 2nd polymer-window (6 µm Mylar), and aluminium lid. The photos (b, c) show the aluminium support (68x 39 mm) and lid (38x 38 mm). The beam window is 3x10 mm wide. |
As depicted in table1, flat window cells are advantageous for structure
studies, mainly because of the thin windows and constant optical pathlenth.
In Fig.6 to 9 some of our flat window cells are shown. The FC1 cells (Fig.6)
for static samples, designed by H.B. Stuhrmann, are easy to assemble but
show a variable background (the polymer windows is asymmetrically stressed
during assembly, the pathlength is not reproducible). Thus they should
be used for strongly scattering samples only. The sample (100 µl)
is inserted into the O-ring space with a adjustable pipette after assembly
of the lower cell half.
A constant optical pathlength is acchieved with the flow-through equipment
(Fig.7 - 9) in the cells FC2 to FC4.
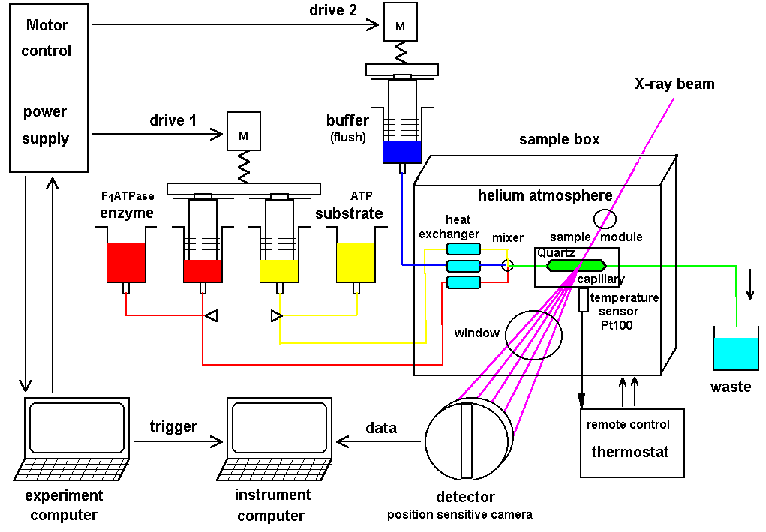 |
| Fig.10: Principal setup for a time resolved X-ray scattering and absorption experiment at a high flux synchrotron. The active working sample is produced from stock solutions by rapid mixing with a stopped-flow device. The helium atmosphere reduces air scattering and beam heating. Radiation demage can be suppressed by slow continous sample displacement by drag buffer with a second drive (from R6, Fig.1). |
The principal setup of a typical X-ray setup for time resolved small
angle scattering is shown in Fig.10 (from
R6).
A picture of this helium cooled sample environment SBox1 and SBox2 for
high flux synchrotron light scattering is shown in Fig.11. The lead shielding
removes background radiation from wiggler and slit sources (if some).
 |
|
Fig.11: Photo of the compact sample environments SBox2 (and SBox1). Component tempering, mixer and flow-through cell are enclosed in a helium filled gas tight box. The sample cell is in SBox2 cooled by a helium jet. The detector side (a) is transparent, the beam side (b) covers a lead shielding. |
After first experiments at ELETTRA, beamline 5.2 (SAXS), a modified
version (SBox2) was equipped with a helium-jet cooling of the sample irradiation
point. Without that cooling an isolated sample (water, 1mm) would develop
gas bubbles and cook after 5 minutes (ELETTRA-SAXS) or 40 seconds (ESRF-ID2A)
irradiation with the full flux. The temperature inside the box is held
by a thermostat regulated with a remote-sensor (Pt100) at the sample module
inside. A sketch of the sample environment SBox2, which was used 1997-1999
at ESRF and ELETTRA, is shown in Fig.12. The environment was used for the
investigation of working F1ATPase and ATP-synthase from Micrococcus
luteus, and in collaboration with H. Heumann and M. Roessle, MPI Martinsried,
for assembly studies of the chaperonin GroEL from Escherichia coli
(see ESRF Newsletter 1999/10, 10).
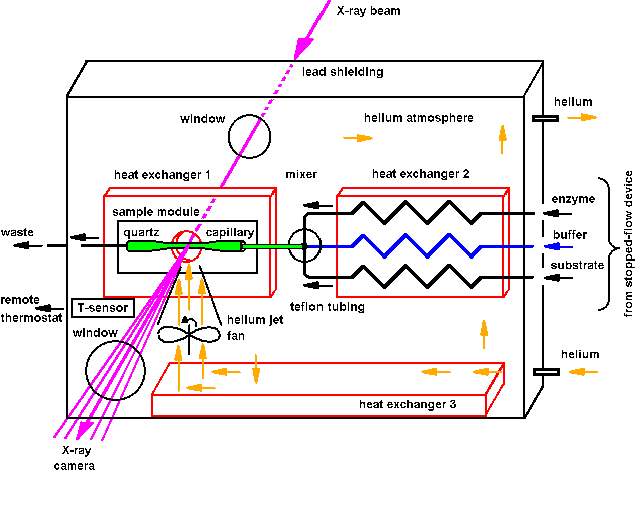 |
| Fig.12: The helium-jet cooled sample environment SBox2 contains a three-branched thermostat, a mixer and the sample cell module in a helium atmosphere for background reduction. Beam heating is reduced by a helium-jet blowing on the capillary lateral to the X-ray beam (< 1 K). The temperature is regulated in a feedback loop with a PT100 sensor at the sample module, which acts on a remote controlled cooling thermostat. After generation of the active sample from protein and substrate stock solutions, the working enzyme can be continously displaced by a slow stream of drag buffer for reduction of the radiation demage. |
Stopped-flow / flash and temperature jump (cold trap double jump) environment : SBox4
A sketch of our recent helium cooled sample environment SBox4 for high
flux synchrotron light scattering is shown in Fig.14. This actual environment,
which is suitable also for temperature jump (cold trap) experiments, is
shown in picture 15. The described double jump technique (double stopped-flow
/ temperature jump = cold trap) was designed for the investigation
of very short structural pulses, which were observed during the reaction
cycle of working F1ATPase.
Equipped with the above flat window flow-through cell, it can be also
used for flash photolysis experiments. This combination of SBox4 was used
at ESRF-ID1 for ASAXS, ESRF-ID24 for time resolved EXAFS, and at DESY-HASYLAB
BW4 (USAXS) for SAXS experiments..
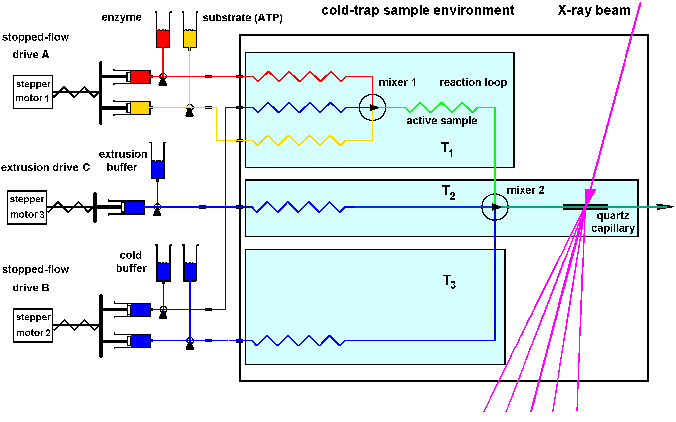 |
| Fig.14: Sketch of a double jump experiment (cold trap) for the investigation of short livetime events using the novel stopped-flow / temperature jump technique: The active sample is in the first experiment phase generated at temperature T1 with stopped-flow drive1, and deposited in a reaction loop. After a delay (computer control) the working complex is pressed out by buffer with the second drive und rapidly mixed with cold buffer of the temperature T2, yielding a temperature jump to T3. The structure is examined at T3 in the capillary module, which is cooled by a helium-jet. Radiation demage can be diminished by slow continous sample displacement by drag buffer usind drive3 (from ESRF application LS1555). |
 |
Fig.15: The modular sample environment SBox4 is constructed for low background high-precission experiments with spectroscopy, X-ray and neutron scattering. The sample cell is cooled by two helium jets. It can be observed with an internal CCD camera. The three thermostats at the right hand side allow temperature jump experiments with the stopped-flow / cold trap method. This enviroment has been used at ESRF Grenoble, beamlines ID1, ID2A and ID24, and at DESY-HASYLAB Hamburg, beamline BW4 (USAXS); a smaller version without temperature jump equipment (SBox2) also at ELETTRA Trieste, beamline 5.2 (SAXS). |
Neutron scattering environments
For time resolved neutron scattering at ILL, beamline D22, we have use
actually a sample environment, which allows rapid-mixing stopped-flow methods
and flash-photolysis techniques as well. As shown in Fig.16, the environment
contains a flat quartz flow-through cuvette (Hellma) and a set of four
UV-flash units in a X-type optics setup. Equipped with UV-filters (305-390
nm) the unit is suitable for the investigation of structure dynamics after
triggering by flash photolysis of caged compounds, e.g. of liposomes during
a pH-jump generated by photolysis of entrapped caged-acid / proton.
| Fig.16: Photo of a sample environment for time resolved neutron scattering as used at ILL Grenoble, beamline D22. The flow-through cell is connevted to a stopped-flow device. The four flash units generate light for photolysis triggered experiments. The X-type optics setup supplies a homogenous distribution of the light inside the sample. |
The control of time resolved experiments and data aquisition is done at the MPSD group by a combination of computer- and digital eletronics (TTL). During this several novel digital and electronics divices were constructed in cooperation with industry. The first experiments in the early 80's where done with timer-chains. Since 1988 we switched over to microcomputer systems (with directly coded binary code for hardware controller CPU's) and TTL-electronics for the fast process steps (microseconds to milliseconds) and PC-computer programs for the slower steps (> 100 ms), providing the parameter and data aquisition / handshake (e.g. F14, T14, T20). An actual example is given in the WEB for time resolved neutron scattering of liposomes (http://www.ill.fr ; see experiment reports ... database-settings: instrument = D22 ; name = Nawroth ; experiment = Test-251 and further). Some technological aspects are presented in the computer control and electronics page.